Eloira - LNG IUS is made of flexible plastic
Eloira contains a progestin hormone called levonorgestrel which has been used in birth control pills since the 1970s
The safety of levonorgestrel has been proven by clinical use also in sub-dermal implants and intrauterine systems since decades
Advantages
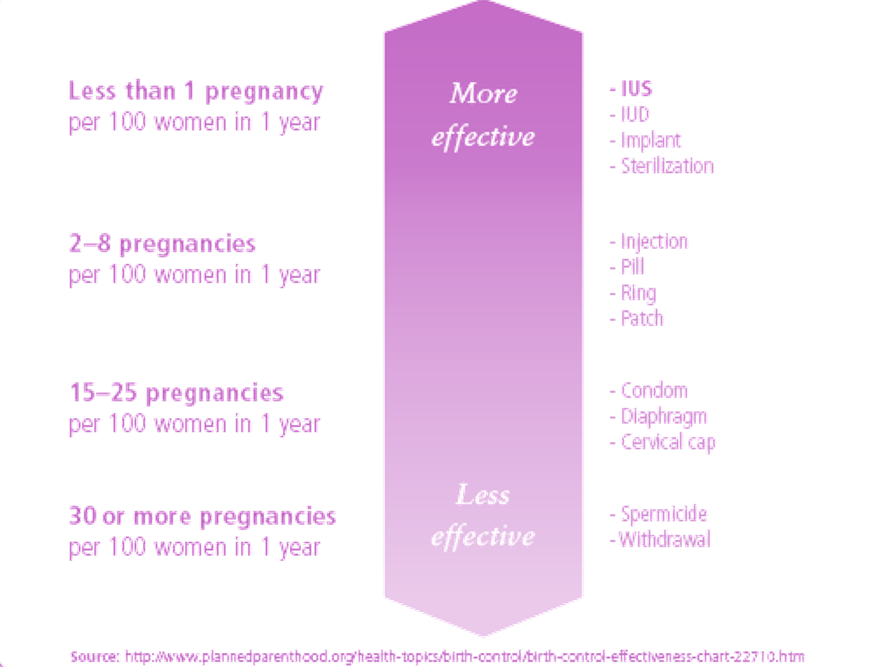
Effective rate of unplanned pregnancy ~0.2% at 1year and less than 1% at 5 years
High continuation rate: up to 85% at one year
Significantly reduces menstrual blood loss
No impact on return to fertility after removal (80% at 1 year)
A woman may choose to use eloira if she:
A woman may choose to use eloira if she:
Wants contraception that provides a low chance of getting pregnant
Wants contraception that is reversible
Wants a contraceptive method that does not require taking it daily
Wants treatment for heavy periods and is willing to use a contraceptive method that is placed in the uterus to prevent pregnancy and a copper IUD would not be appropriate
Indications for use
Contraception
Dysfunctional Uterine Bleeding
Endometrial Hyperplasia
Fibroids
Adenomyosis
Endometriosis
HRT
Alternative to Sterilization
Where sterilization services are not available or not acceptable, IUS is as effective as sterilization (8 vs. 13 failures per 1000 procedures at 5 years)
Reversibility of LNG IUS reduces risk of regret
Complementing female sterilization in northern Europe
Treatment for HMB
Use of LNG IUS makes periods lighter, shorter and less painful (and may include dysmenorrhea)
Over 12 months, blood loss reduced by 80-96% in women with menorrhagia
Clinical improvement in associated anemia: Hemoglobin levels rise 1.8g/L in one year of use with LNG IUS, compared to a decrease of 1.2g/L with Copper-T.
Irregular bleeding or spotting common in first 3-6 months; 20% with amenorrhea at 12 months. Sometimes the amenorrhea rate can be higher than 20% e.g. up to 50% at 12 and 24 months of use